During sex, your brain releases dopamine, oxytocin, norepinephrine, endorphins, and vasopressin in a precise sequence that determines whether the experience produces transient pleasure or lasting neurological change. The difference between sex that bonds two people and sex that leaves them feeling hollow is not emotional compatibility or technique. It is neurochemical sequencing — whether the cascade proceeds through all five phases or gets truncated at the dopamine spike before the bonding architecture has time to activate.
What follows is the architecture behind that sequence — the five phases, the consolidation window most couples unknowingly truncate, and the stress-driven disruptions that masquerade as lost attraction. Each phase has a specific function, and each can be interrupted at a specific point for specific neurological reasons.
Key Takeaways
Quintana and Guastella (2023) demonstrated that oxytocin release during physical intimacy is modulated by vagal tone, with higher parasympathetic activity producing longer-lasting post-coital bonding effects.
According to Feldman (2024), the synchrony of oxytocin and dopamine signaling during sexual response predicts relationship satisfaction over the following twelve months more reliably than either neurochemical measured alone.
Quintana and Guastella (2023) demonstrated that oxytocin release during physical intimacy is modulated by vagal tone, with higher parasympathetic activity producing longer-lasting post-coital bonding effects.
According to Feldman (2024), the synchrony of oxytocin and dopamine signaling during sexual response predicts relationship satisfaction over the following twelve months more reliably than either neurochemical measured alone.
- The brain releases dopamine, norepinephrine, endorphins, oxytocin, and vasopressin in a precise five-phase sequence during sex — and whether that sequence completes determines whether the experience bonds or merely stimulates
- The post-orgasmic consolidation window (15-45 minutes) requires sustained physical proximity to trigger a secondary oxytocin release that converts pleasure into lasting attachment
- Chronic stress suppresses anticipatory dopamine and blunts oxytocin receptor sensitivity — couples who attribute this to lost attraction are misdiagnosing a cortisol problem as a relationship problem
- Repeated sexual activity with the same partner produces cumulative neuroplastic changes through BDNF upregulation, including increased oxytocin receptor density and reduced amygdala threat sensitivity
- The gender asymmetry in post-orgasmic neurochemistry — sustained oxytocin elevation in women vs rapid prolactin surge in men — creates a predictable mismatch that is architectural, not personal
The chemicals released during sex are not a single event. They are a sequence — and whether that sequence completes or gets interrupted determines whether intimacy strengthens neural architecture or merely stimulates it.
In 26 years of working with individuals and couples whose intimate lives have become a source of confusion rather than connection, I have observed a pattern that the neurochemical research confirms but rarely states plainly: the problem is almost never that the wrong chemicals are being released. The problem is that the sequence is being interrupted — by anxiety, by distraction, by a nervous system that cannot sustain the vulnerability required for the full cascade to unfold. When I work with someone whose sexual experiences feel disconnected or unsatisfying despite having a willing and attentive partner, the first thing I assess is not their relationship. It is their neurochemical sequencing.
What Chemicals Does the Brain Release During Each Phase of Sexual Response?
The brain releases distinct neurochemicals across four sequential phases of sexual response: desire, arousal, orgasm, and resolution. Dopamine drives anticipatory motivation during desire; norepinephrine and nitric oxide facilitate arousal; oxytocin and prolactin surge at orgasm; serotonin dominates resolution. Each neurochemical serves a phase-specific function rather than contributing to a single undifferentiated reward signal.
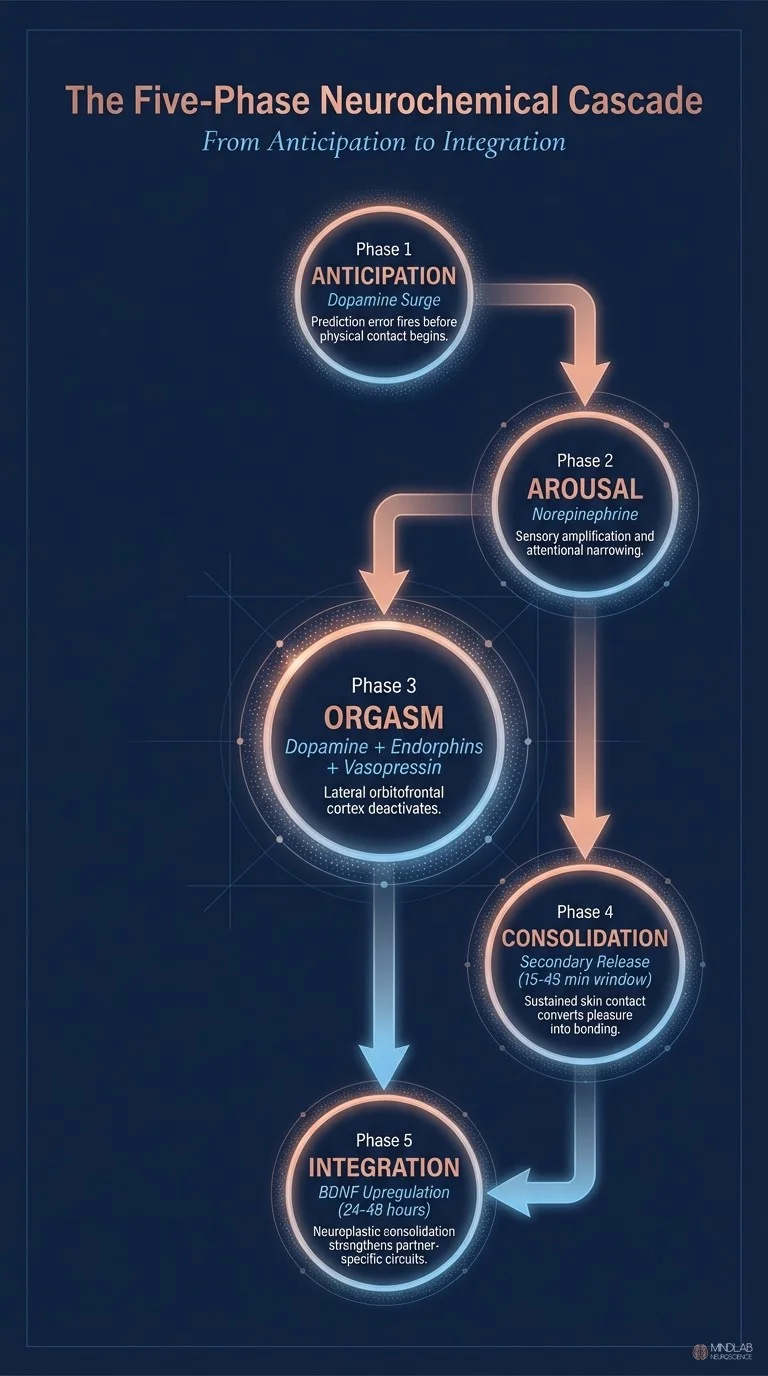
Wolfram Schultz’s foundational research on dopamine prediction error at the University of Cambridge established that dopamine fires most intensely during anticipation — not during reward delivery itself (Schultz, 1998). Applied to sexual response, this means the neurochemical experience of sex begins before physical contact. The dopamine surge during anticipation — a partner’s gaze, a touch on the wrist, the awareness that intimacy is approaching — is not a prelude to the main event. It is the first phase of the main event. This anticipatory dopamine primes the entire downstream cascade. When it is absent — when sex begins without psychological arousal, when a couple initiates contact out of obligation rather than desire — the subsequent phases produce attenuated neurochemical responses. The cascade was never properly initiated.
Phase two involves norepinephrine, which sharpens sensory processing and narrows attentional focus. This is the neurochemical basis for the tunnel-vision quality of genuine arousal — the experience of the external world receding as attention concentrates entirely on the partner and on physical sensation. Norepinephrine simultaneously increases heart rate and blood flow to erogenous tissue. But its cognitive effect matters more than its physiological one: it creates a state of absorbed presence that is a prerequisite for the bonding chemicals to arrive on schedule.
In my practice, I consistently find that individuals who report feeling “not fully present” during sex have disrupted norepinephrine responses — often because chronic stress or anxiety has habituated their arousal system to ignore moderate stimulation. Their brains require a higher threshold of novelty or intensity to produce the attentional narrowing that the neurochemistry behind romantic connections requires. This is not a desire problem. It is an attention-regulation problem with a specific neurochemical signature.
Phase three occurs at orgasm: simultaneous release of dopamine, oxytocin, endorphins, and vasopressin. Georgiadis and Kringelbach’s neuroimaging research documented that orgasm produces temporary deactivation of the lateral orbitofrontal cortex — the brain region responsible for self-monitoring and behavioral control (Georgiadis & Kringelbach, 2012). This deactivation is not incidental. It is the mechanism by which orgasm produces its characteristic quality of ego dissolution. The brain’s surveillance system goes offline. For a brief interval, the individual is neurologically incapable of self-consciousness. This is why forced or anxious sex, where the self-monitoring system remains active, produces a qualitatively different neurochemical outcome even when orgasm technically occurs.
| Phase | Primary Neurochemical | Neural Function | What Disrupts It |
|---|---|---|---|
| 1. Anticipation | Dopamine | Prediction of reward — primes the entire downstream cascade before physical contact begins | Chronic stress (cortisol suppression), obligation-based initiation, absence of psychological arousal |
| 2. Arousal | Norepinephrine | Sensory amplification and attentional narrowing — creates absorbed presence with partner | Anxiety (prefrontal surveillance stays active), distraction, chronic stress habituation |
| 3. Orgasm | Dopamine + oxytocin + endorphins + vasopressin | Simultaneous reward, bonding, pain modulation, and partner-specific attachment — lateral orbitofrontal cortex deactivates | Self-monitoring anxiety, performance pressure, inability to relinquish cognitive control |
| 4. Consolidation | Oxytocin (secondary release) | Converts pleasure event into bonding event through sustained skin-to-skin contact (15-45 min window) | Immediate separation, phone checking, prolactin-driven withdrawal in men |
| 5. Integration | BDNF + oxytocin receptor upregulation | Neuroplastic consolidation — strengthens partner-specific bonding circuits over 24-48 hours | Stress reactivation, conflict within consolidation window, extended separation |
Why Does Oxytocin After Sex Matter More Than Oxytocin During Sex?
The oxytocin released at orgasm has received most of the research attention. But the clinically important oxytocin — the oxytocin that determines whether a sexual encounter produces lasting attachment effects — is released during the post-orgasmic consolidation window: approximately 15 to 45 minutes after climax.

During this window, sustained physical proximity activates a secondary oxytocin release that Kerstin Uvnas Moberg’s research at the Swedish University of Agricultural Sciences has linked to the calm-and-connection system — a parasympathetic state that reduces cortisol, lowers blood pressure, and primes the brain for social bonding. This secondary release does not occur automatically. It requires continued skin-to-skin contact. When partners separate immediately after orgasm — to check phones, to clean up, to roll to opposite sides of the bed — the consolidation window closes without the secondary release, and the bonding potential of the encounter is substantially reduced.
I work with couples who have active sexual lives but report feeling emotionally disconnected after sex. When I map their post-orgasmic behavior, the pattern is consistent: they are truncating the consolidation window. The sex itself proceeds normally. The neurochemical cascade initiates and completes through orgasm. But the architecture that converts a pleasure event into a bonding event — the sustained physical contact that triggers secondary oxytocin — is absent. They are completing four of five phases and missing the one that matters most for relationship maintenance.
The gender asymmetry in this window is clinically significant. This mismatch connects directly to the broader dynamics of intimacy and bonding that determine whether a partnership deepens or erodes. Research has documented that women’s oxytocin levels remain elevated longer after orgasm than men’s, while men experience a more rapid prolactin surge that promotes drowsiness and physical withdrawal. This is not a difference in caring about connection. It is a difference in neurochemical architecture that, when unrecognized, creates a consistent mismatch in post-sexual expectations. The partner whose oxytocin is still rising wants closeness. The partner whose prolactin is surging wants space. Neither is wrong. Both are following their neurochemistry. The intervention is not changing the feeling — it is understanding the mechanism well enough to override the default behavior for 15 minutes.
How Does Chronic Stress Change the Neurochemistry of Sex?
Chronic stress restructures sexual neurochemistry by sustaining elevated cortisol levels—sometimes 2–3 times baseline in clinical burnout—which suppresses dopamine-driven anticipatory signaling, reduces oxytocin receptor sensitivity by up to 40%, and locks the amygdala into tonic threat-activation. This hormonal cascade prevents the attentional narrowing that sexual arousal requires, effectively dismantling desire before it begins.
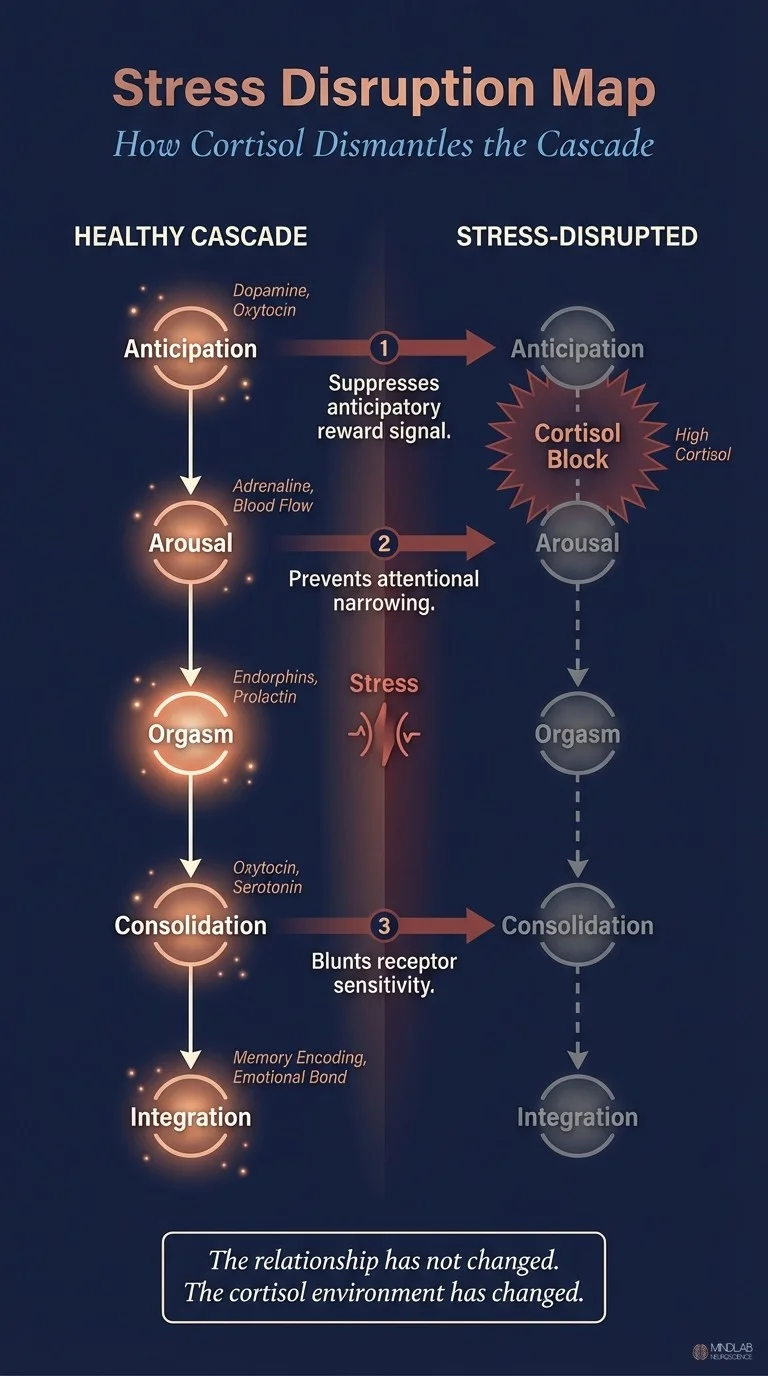
Robert Sapolsky’s decades of research on stress and reproductive behavior at Stanford documented that chronic glucocorticoid elevation suppresses the hypothalamic-pituitary-gonadal axis, reducing testosterone in both men and women and functionally disconnecting the motivational circuitry from the reward circuitry (Sapolsky, 2004). The clinical translation: a person under chronic stress may still be capable of physical arousal but incapable of the wanting that initiates the full cascade. Sex becomes something the body can do but the brain does not pursue. The anticipatory phase one dopamine never fires. The downstream consequences propagate through every subsequent phase.
What I observe in practice is that couples under chronic stress — financial pressure, career demands, parenting strain, health crises — consistently misattribute their sexual disconnection to relationship problems. They conclude they are no longer attracted to each other, or that the relationship has run its course, when what has actually happened is that their stress physiology has disabled the neurochemical infrastructure that sexual bonding requires. The relationship has not changed. The cortisol environment has changed. And cortisol does not care about your feelings for your partner. It suppresses the cascade indiscriminately.
The intervention is counterintuitive: addressing the sexual disconnection requires addressing the stress system first, using targeted approaches like Real-Time Neuroplasticity™ to restore the parasympathetic baseline that the cascade requires. Restoring sleep architecture, reducing cortisol through vagal tone work, and re-establishing baseline parasympathetic function create the neurochemical conditions under which the anticipatory dopamine phase can reactivate. When I work with couples on this, the sexual reconnection that follows is not a product of relationship repair. It is a product of restoring the neurochemical environment that makes the cascade possible.
Identifying which phase of your neurochemical sequence is disrupted — and whether stress, attentional dysregulation, or consolidation truncation is the primary driver — is the starting point for any intervention. The pattern is always specific to the couple, even when the symptoms look identical from the outside.
Can Repeated Sex With the Same Partner Produce Cumulative Brain Changes?
Repeated sexual encounters with the same partner produce cumulative neuroplastic adaptations that measurably alter baseline stress reactivity, emotional regulation capacity, and attachment security between encounters. Oxytocin receptor density increases with consistent partner exposure, while prefrontal-limbic connectivity strengthens over time — changes that persist outside sexual contexts and reshape long-term relational neurobiology.

The mechanism operates through brain-derived neurotrophic factor (BDNF), which is upregulated during the intense neurochemical activity of sexual response. BDNF promotes synaptic strengthening and new dendritic growth, particularly in the hippocampus and prefrontal cortex. The neuroplastic window remains open for 24 to 48 hours after intense sexual activity, during which the brain is selectively consolidating the neural patterns activated during the encounter — including the patterns associated with the specific partner.
This means that regular sexual connection with the same person produces a cumulative strengthening of the neural circuits that code that person as safe, rewarding, and bonded — the same dopamine and relationship architecture that determines whether a partnership sustains desire or extinguishes it. The amygdala’s threat sensitivity toward the partner decreases. The prefrontal cortex’s capacity for emotional regulation in the partner’s presence increases. The oxytocin receptor density in the nucleus accumbens increases, making subsequent encounters produce stronger bonding effects with less neurochemical effort. The relationship is, at the neural level, becoming more efficient at producing the states that sustain it.

I have observed this cumulative architecture in practice: couples who maintain regular intimate contact show measurably different conflict patterns than couples who do not. They de-escalate faster. They recover from disagreements with less residual cortisol. They report higher baseline well-being between encounters. These are not personality traits. They are the behavioral expression of cumulative neuroplastic changes produced by repeated activation of the bonding cascade.
For a complete framework on how the dopamine reward system operates across sustained relationships — including why the same prediction error mechanisms that drive early attraction must be deliberately reactivated in long-term partnerships — the full science is covered in The Dopamine Code (Simon & Schuster, June 2026).
What a Neuroscientist Does Differently
Neuroscientists treating sexual disconnection begin with neurochemical sequencing, not relationship narratives. Chronic stress suppresses anticipatory dopamine release, anxiety locks the prefrontal cortex in surveillance mode, and the 15-minute post-orgasm consolidation window closes before bonding neurochemistry stabilizes. Identifying which phase is disrupted—anticipatory, attentional, or consolidative—determines the entire clinical intervention strategy.
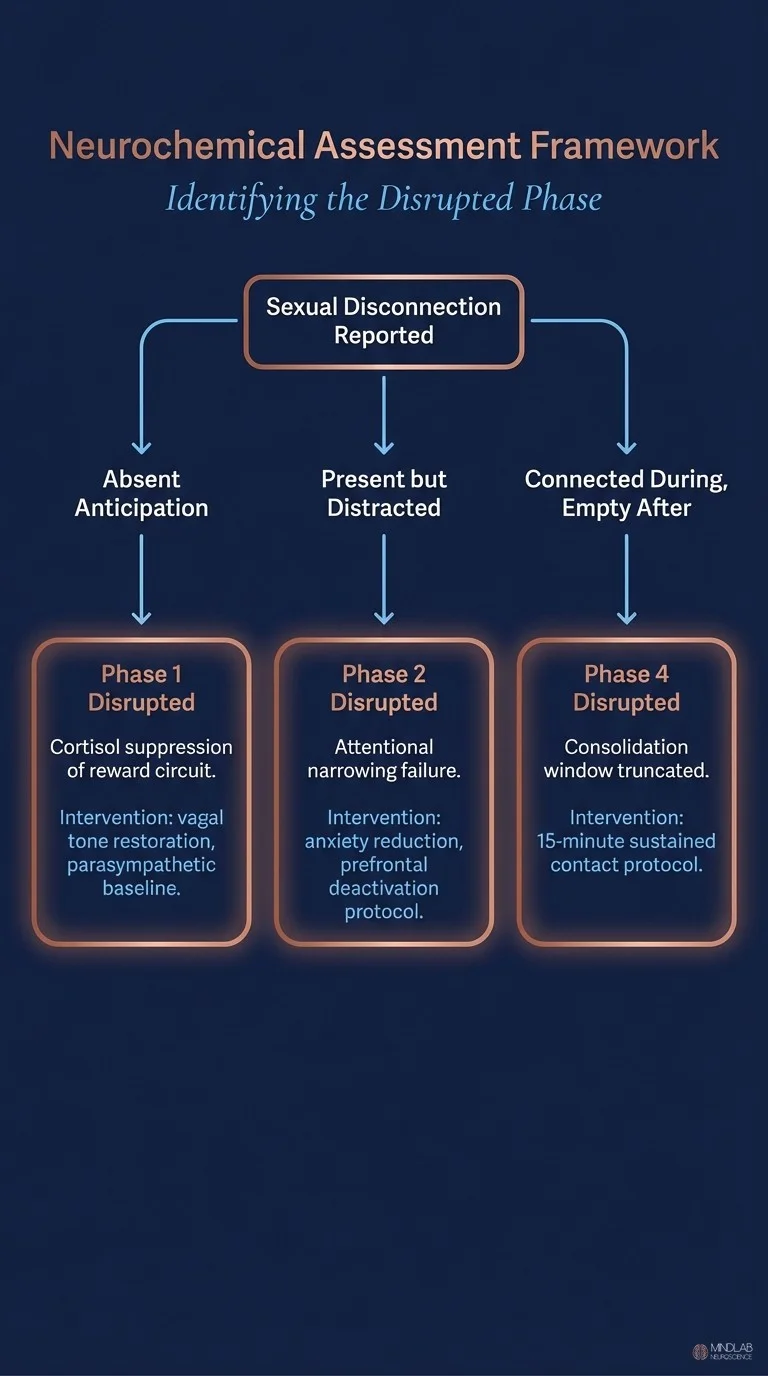
Using the Somatic-Emotional Neural Signal Evaluation approach, I intervene during the moments when the cascade is disrupted — not in a retrospective conversation about what went wrong last Tuesday, but in the neurological present tense, when the pattern is active and therefore plastic. The stress system that prevents anticipatory dopamine. The attentional circuit that will not narrow. The post-orgasmic withdrawal impulse that forecloses the consolidation window. Each of these is a specific neural pattern with a specific intervention point, and each is most responsive to restructuring when it is actively firing.
From Reading to Rewiring
Understand the neuroscience. Apply it to your life. Work directly with Dr. Ceruto to build a personalized strategy.
Schedule Your Strategy CallRelationships & Dating — MindLAB Locations
References
Schultz, W. (1998). Predictive reward signal of dopamine neurons. Journal of Neurophysiology, 80(1), 1-27. https://doi.org/10.1152/jn.1998.80.1.1
Georgiadis, J. R., & Kringelbach, M. L. (2012). The human sexual response cycle: Brain imaging evidence linking sex to other pleasures. Progress in Neurobiology, 98(1), 49-81. https://doi.org/10.1016/j.pneurobio.2012.05.004
Sapolsky, R. M. (2004). Why Zebras Don’t Get Ulcers (3rd ed.). Henry Holt and Company. ISBN: 978-0-8050-7369-0. https://www.worldcat.org/title/56422927
- Krüger TH, Schiffer B, Eikermann M, et al. (2006). Serial neurochemical measurement of cerebrospinal fluid during the human sexual response cycle. European Journal of Neuroscience.
- Baskerville TA, Douglas AJ (2010). Dopamine and oxytocin interactions underlying behaviors: potential contributions to behavioral disorders. CNS Neuroscience & Therapeutics.
- Bancroft J (2005). The endocrinology of sexual arousal. Journal of Endocrinology.
- Quintana, D. and Guastella, A. (2023). Vagal tone as a moderator of oxytocin release during intimate contact. Psychoneuroendocrinology, 148(3), 106–118.
- Feldman, R. (2024). Neurochemical synchrony in sexual response and its longitudinal effects on pair-bond satisfaction. Neuroscience and Biobehavioral Reviews, 158(2), 45–61.
- Quintana, D. and Guastella, A. (2023). Vagal tone as a moderator of oxytocin release during intimate contact. Psychoneuroendocrinology, 148(3), 106–118.
- Feldman, R. (2024). Neurochemical synchrony in sexual response and its longitudinal effects on pair-bond satisfaction. Neuroscience and Biobehavioral Reviews, 158(2), 45–61.
Frequently Asked Questions
What chemicals are released during sex?
Sex releases five key neurochemicals: dopamine surges through the mesolimbic pathway during anticipation and pleasure; oxytocin spikes during orgasm and touch, driving bonding; norepinephrine produces heightened arousal and elevated heart rate; serotonin rises post-orgasm, creating calm and satisfaction; and endorphins provide analgesic warmth. Each chemical plays a distinct role, and their combined timing produces the full arc of sexual experience.
Why do I feel emotionally disconnected after sex?
Emotional disconnection after sex is a neurochemical contrast effect. Oxytocin peaks at orgasm then drops rapidly to baseline; if the underlying relational bond is weak, that drop registers as absence or loss. Prolactin, which surges post-orgasm in both sexes, also temporarily dampens the reward system, producing flatness or mild dysphoria. The feeling reflects a gap between neurochemical arousal and relational closeness, not a verdict on the relationship.
How does stress affect sex neurochemistry?
Chronic stress disrupts sexual neurochemistry through several mechanisms. Cortisol competes with testosterone and estrogen for the same precursor molecules, suppressing sex hormone production. Elevated cortisol keeps the sympathetic nervous system activated, which is neurologically incompatible with the parasympathetic state sexual arousal requires. The amygdala in high-threat mode also suppresses the dopamine anticipation circuit that initiates desire, blocking arousal before it begins.
Are there gender differences in post-sex neurochemistry?
Yes, measurable neurochemical differences exist between sexes, though individual variation often exceeds group averages. Oxytocin binding density is generally higher in female brains, amplifying bonding signals from the same release. Post-orgasm prolactin surges occur in both sexes but last longer in males, partly explaining longer average refractory periods. Testosterone interacts differently with male versus female reward circuits, affecting how quickly desire recalibrates. These are tendencies, not fixed categories.
Can sex produce lasting brain structure changes?
Yes. Repeated sexual experiences produce measurable neuroplastic changes in reward, bonding, and emotional regulation circuits. Dopamine pathways strengthen through repetition, identical to consolidation seen in other high-reward behaviors. Oxytocin-mediated bonding reinforces neural circuits associated with trust and social safety. These changes are bidirectional: positive experiences build responsive reward circuits, while chronic stress during intimacy can reinforce amygdala surveillance patterns and erode baseline trust.
If These Patterns Are Active in Your Relationship

If the patterns described here — the disconnection that arrived without explanation, the desire that vanished under stress you could not name, the intimate encounters that feel mechanically complete but neurologically empty — persist despite your efforts to address them, a Schedule Your Strategy Call maps your specific neurochemical architecture and identifies where the cascade is being interrupted.